I've been reading through the thread and I kept seeing things like this.
brigit wrote:
Hi Lloyd,
As you pointed out, for most of the electrical transmutation to work in the fossilization process, you need some added protons (or neutrons or whichever) to get to the silica or calcium or phosphorous, or iron. I am all for balancing the equations. But first, it seems to me, if I remember correctly, that the numbers given in the periodic tables are averages, so if you have the right isotopes of an element, you can get to silica more directly, or the Iridium from Iron. Next, the additional nuclear material needed to change one element to another could come from the air, without the need for the water.
You don't need to have an external source of protons or neutrons to work the transmutation, just add energy to an existing molecule. Everything happens inside.
Keep in mind that the atom is more like a bunch of dancing m&m candies--holding hands, or expanding to swallow each other like nested russian dolls--than billiard balls, and all of this will make sense.
When you think transmutation, remember that everything is in some molecular bond. That bond brings the nucleus of the atoms closer than what you remember from high school chemistry class.
Remember high school chemistry class, we played with tinkertoy like models; colored balls connected by sticks. It was a simple way to visualize what was happening but it creates a category error when we look at the real world. In reality the atoms are embedded in a common electron shell and can trade protons and neutrons under the right conditions.
In other words, atoms in molecules don't look like tinkertoys connected with sticks, they look like this:

- 180px-Nitrogen-3D-vdW.png (11.89 KiB) Viewed 23070 times
N
2
And this:

- 120px-Carbon-monoxide-3D-vdW.png (5.77 KiB) Viewed 23071 times
CO, carbon monoxide
At this level the molecule acts like a single atom, trying to fill its electron shell. (Pull up the page for
Electron shells and have it sitting in another window so you can check back and forth. (You might want to read through it to get an idea of the concept; it's a short page, but key to many things.))
- Atoms join in chemical bonds to fill their electron shells. They want to complete their electron shells, to be stable.
Look at
List of elements with electrons per shell
The Nitrogen molecule, N
2, basically wants its electron shells to fill up and act like a single atom.
This is N
2 from the table:
Nitrogen 2, 5
Nitrogen 2, 5
It breaks out to this:
Nitrogen 2, 2+3=5
Nitrogen 2, 2+3=5
And looks like this by shell and subshell:
Nitrogen K s 2, L s 2 + L p 3
Nitrogen K s 2, L s 2 + L p 3
Look at it using parentheses for the {shells} and (subshells) and |atom|.
The inner K-shell looks like this.
Nitrogen |{(({K s 2} L s 2) L p 3)}|
The inner K-shell is contained in the L-shell, s-subshell.
Nitrogen |{(({K s 2} L s 2) L p 3)}|
Then everything is within the (L p) subshell.
Nitrogen |{(({K s 2} L s 2) L p 3)}|
So the Nitrogen molecule would start with two Nitrogen atoms:
Nitrogen |{(({K s 2} L s 2) L p 3)}|
Nitrogen |{(({K s 2} L s 2) L p 3)}|
The Nitrogen molecule has two first and second s subshells filled, and they each have half their p subshells filled. The molecule likes sharing the p subshell because that fills it. It acts as if it were one atom with the (L p 6) filled. It's those six electrons that wrap up the entire Nitrogen molecule making it chemically stable, that's why they store documents like the Declaration of Independence in a N
2 gas filled container. The molecule acts like a Noble gas, in this case Neon.
Neon |{(({K s 2} L s 2) L p 6)}|.
Look at the two Nitrogen atoms with their electron shells spread out. See how the outer (L p) subshell wraps around both atoms and is filled with six electrons. Which means the whole molecule acts like it is an atom, which is why the bars "|" have been placed around the whole
Nitrogen molecule, N
2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
The inner K-shells looks like this.
Nitrogen molecule, N2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
The inner K-shells are contained in the L-shell, s-subshell.
Nitrogen molecule, N2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
Then everything is within the (L p) subshell.
Nitrogen molecule, N2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
That's why the Nitrogen molecule looks like this.

- 180px-Nitrogen-3D-vdW.png (11.89 KiB) Viewed 23070 times
N
2
_____________
Now here is where things get strange:
- A Neutron lasts for only 15 minutes before it breaks down into a Proton, Electron, and Neutrino.
Keep something very important in mind:
- Even in the heart of the nucleus, the neutron last only 15 minutes.
Now this is where I get into trouble. When you read the Wiki pages they won't mention that. I've seen videos of the proton neutron switch many times on PBS, but I can't find a reference or the videos, so take this part with a large grain of salt until a solid reference is found.
Here's where the dancing m&m candies comes in to play.
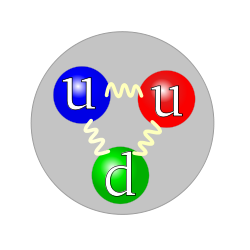
- 250px-Quark_structure_proton.svg.png (19.82 KiB) Viewed 23067 times
Proton
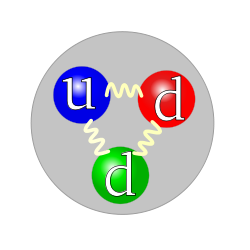
- 250px-Quark_structure_neutron.svg.png (20.15 KiB) Viewed 23066 times
Neutron
See how the only difference between the proton and the neutron is the red m&m changing from an up quark to a down quark. That's where the action is happening. Always remember that when you look at the picture of the dancing m&ms, there are not just three m&ms, there are other m&ms dancing around and inside acting as the forces--called strong and weak--that hold them together in their candy coated shell.
Now look at the simple example of Deuterium, which is an isotope of Hydrogen. The nucleus is a proton and a neutron, with one electron orbiting the whole. Every 15 minutes, the neutron decays to a proton-electron-neutrino and the proton becomes the neutron by absorbing the electron-neutrino from the decaying neutron, all the while the outer electron orbits.
There is no electron actually orbiting around the nucleus, the electron surrounds the nucleus in a shell. The nucleus is nested within the electron.
Put simply the red m&ms change up and down by having other m&ms swap back and forth, while the electron m&m wraps the nucleus inside a candy coated shell while other m&ms hold everything together.
This is the family tree of the various m&ms, all nested and dancing around and within each other.
Standard model of elementary particles
- Think about that simple example and the massive implications of it, because this is the engine that drives the transmutation.
_____________
Let's look at a couple of molecules and follow them through a transmutation based on their electron shells.
_____________
The Nitrogen molecule mentioned above is stable, calmly being N
2. The thing about the electron shells, is that the electrons in each outer shell want to fall to the lowest energy possible, so when they get hit with photons of the right frequency they absorb energy and change their subshell.
- An important point, the electrons in a subshell have exactly the same level of energy, so when energy pumps into the (L p 6) subshell the whole subshell changes as one.
Left on their own, the electrons will dump the energy as a photon and fall back to the stable structure, but as mentioned far back in the thread biological interaction with the N
2 molecule can change it from N
2 to carbon monoxide, CO.
As you may remember, welders would fall sick from carbon monoxide poisoning yet air detectors would not see carbon monoxide around them, it only showed up in the blood. Yet clearly N
2 was becoming CO when absorbed by the lungs.
If you look at the
List of elements with electrons per shell, you can see how Carbon, Nitrogen, and Oxygen relate.
6 Carbon 2, 4
7 Nitrogen 2, 5
8 Oxygen 2, 6
7 Nitrogen 2, 5
7 Nitrogen 2, 5
Total 14 proton/neutrons
Becomes:
6 Carbon 2, 4
8 Oxygen 2, 6
Total 14 proton/neutrons
We can break out the {shells} and (subshells) like before:
Carbon |{(({K s 2} L s 2) L p 2)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
So:
Nitrogen molecule, N
2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
Becomes:
Carbon monoxide, CO, |{((L s 2 {K s 2})Carbon|Oxygen({K s 2} L s 2) L p 6)}|
The inner K-shells looks like this.
Carbon monoxide, CO, |{((L s 2 {K s 2})Carbon|Oxygen({K s 2} L s 2) L p 6)}|
The inner K-shells are contained in the L-shell, s-subshell.
Carbon monoxide, CO, |{((L s 2 {K s 2})Carbon|Oxygen({K s 2} L s 2) L p 6)}|
Then everything is within the (L p) subshell.
Carbon monoxide, CO, |{((L s 2 {K s 2})Carbon|Oxygen({K s 2} L s 2) L p 6)}|
Notice, CO still has the outer (L p 6) subshell filled.
The way this transformation occurs is when the N
2 is absorbed by the lungs it becomes part of a larger molecule while still energized by the red hot metal and doesn't have the chance to lose energy by dumping a photon, and in the random play of protons and neutrons dancing in the nucleus--with the subshell disrupted--that energy has to go somewhere, so the nucleus takes the hit.
- In other words, all of the dancing m&ms get energized and do the unexpected.
This is like radioactive decay, it is random, and only a percentage of the excited N
2 will transmute to CO, but a neutron and proton will end up moving from one nitrogen atom to the other; and what's really crazy is that they look like they jumped from one nucleus to the other without traveling the space in between. That of course seems impossible until you remember that electrons "jump" from one shell or subshell to the next instantly, without crossing in between.
_____________
Now lets look at the Oxygen molecule, O
2 and see how it changes to Sulphur.
O
2
8 Oxygen 2, 6
8 Oxygen 2, 6
Total 16 proton/neutrons
16 Sulfur 2, 8, 6
Total 16 proton/neutrons
Oxygen |{(({K s 2} L s 2) L p 4)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
Notice, that each Oxygen atom (L p) subshell is already almost filled. When the two Oxygen atoms come together an interesting thing happens, the (L p) subshells come together, but it can't be (L p 8) it can only be (L p 6) which leaves two electrons to form a new shell around the whole molecule (M s 2).
Oxygen molecule, O
2, |{({((L s 2 {K s 2})|({K s 2} L s 2) L p 6)} M s 2)}|
The inner K-shells looks like this.
Oxygen molecule, O2, |{({((L s 2 {K s 2})|({K s 2} L s 2) L p 6)} M s 2)}|
The inner K-shells are contained in the L-shell, s-subshell.
Oxygen molecule, O2, |{({((L s 2 {K s 2})|({K s 2} L s 2) L p 6)} M s 2)}|
Then everything is within the (L p) subshell.
Oxygen molecule, O2, |{({((L s 2 {K s 2})|({K s 2} L s 2) L p 6)} M s 2)}|
Then everything is within the M-shell.
Oxygen molecule, O2, |{({((L s 2 {K s 2})|({K s 2} L s 2) L p 6)} M s 2)}|
This means the two oxygen atoms are embedded together within the electron L-shell and the M-shell. Electrons in the M-shell are a different shape and energy than those in the L-shell.
When the oxygen molecule is hit with a massive burst of energy the nucleus are fused together by everyones favorite z-pinch to make ionized sulfur. That ionized sulphur atom reacts with the surrounding oxygen to form sulphur dioxide.
- 100px-Sulfur-dioxide-3D-vdW.png (5.68 KiB) Viewed 23072 times
Sulfur dioxide, SO
2
Sulfur |{(({(({K s 2} L s 2) L p 6)} M s 2) M p 4)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
Sulfur dioxide, SO
2, |{({(L p 6 (L s 2 {K s 2}))}Oxygen|Sulfur{(({K s 2} L s 2) L p 6)}Sulfur|Oxygen{(({K s 2} L s 2) L p 6)}Oxygen|Sulfur M s 2)}|
The inner K-shells looks like this.
Sulfur dioxide, SO2, |{({(L p 6 (L s 2 {K s 2}))}Oxygen|Sulfur{(({K s 2} L s 2) L p 6)}Sulfur|Oxygen{(({K s 2} L s 2) L p 6)}Oxygen|Sulfur M s 2)}|
The inner K-shells are contained in the L-shell, s-subshell.
Sulfur dioxide, SO2, |{({(L p 6 (L s 2 {K s 2}))}Oxygen|Sulfur{(({K s 2} L s 2) L p 6)}Sulfur|Oxygen{(({K s 2} L s 2) L p 6)}Oxygen|Sulfur M s 2)}|
Then everything is within the (L p) subshell.
Sulfur dioxide, SO2, |{({(L p 6 (L s 2 {K s 2}))}Oxygen|Sulfur{(({K s 2} L s 2) L p 6)}Sulfur|Oxygen{(({K s 2} L s 2) L p 6)}Oxygen|Sulfur M s 2)}|
Then everything is within the M-shell.
Sulfur dioxide, SO2, |{({(L p 6 (L s 2 {K s 2}))}Oxygen|Sulfur{(({K s 2} L s 2) L p 6)}Sulfur|Oxygen{(({K s 2} L s 2) L p 6)}Oxygen|Sulfur M s 2)}|
Notice that the two oxygen atoms want to fill their outer (L p) subshell and they get the four electrons they need from the outer (M p 4) subshell of the sulfur atom. The (L p) subshells of the oxygen do not connect with the sulfur (L p) subshell, each is completely filled. The (M s 2) subshell is what surrounds everything, but the four electrons from the (M p 4) subshell of the sulfur atom still "belong" to the sulfur.
If you look at the picture of the Sulfur dioxide molecule, you can see the oxygen atoms are close to each other. That may be based on the way the different subshells orbit. Look at the
Orbital table and remember that three different geometries come together to form the final molecule. All the various forces shape and tug the molecule, but it still wants to act like a single atom with an outer (M s 2) subshell.
_____________
Now lets look at how the Nitrogen molecule, N
2, can become Silicon, and ultimately Silicon dioxide, SiO
2.
7 Nitrogen 2, 5
7 Nitrogen 2, 5
Total 14 proton/neutrons
14 Silicon 2, 8, 4
Total 14 proton/neutrons
In the example above we could see how the Nitrogen molecule looks as far as the electron shells go.
Nitrogen molecule, N
2, |{((L s 2 {K s 2})|({K s 2} L s 2) L p 6)}|
When a lightning bolt hits, the same Z-pinch effect that crunched the Oxygen molecule into Sulfur can crunch the Nitrogen molecule into Silicon.
- With all that energy being pumped in, the m&ms go wild. A number of elements would be fused, transmuted, ionized, etc..., all in the same bolt of lightning.
And the Silicon ion joins with Oxygen to make Silicon dioxide, so we can keep Brigit happy.
Silicon dioxide, SiO
2
Silicon |{(({(({K s 2} L s 2) L p 6)} M s 2) M p 2)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
Oxygen |{(({K s 2} L s 2) L p 4)}|
Silicon dioxide, SiO
2, |{(L p 6 (L s 2 {K s 2}))}Oxygen|Silicon{(({K s 2} L s 2) L p 6)}Silicon|Oxygen{(({K s 2} L s 2) L p 6)}|
The inner K-shells looks like this.
Silicon dioxide, SiO2, |{(L p 6 (L s 2 {K s 2}))}Oxygen|Silicon{(({K s 2} L s 2) L p 6)}Silicon|Oxygen{(({K s 2} L s 2) L p 6)}|
The inner K-shells are contained in the L-shell, s-subshell.
Silicon dioxide, SiO2, |{(L p 6 (L s 2 {K s 2}))}Oxygen|Silicon{(({K s 2} L s 2) L p 6)}Silicon|Oxygen{(({K s 2} L s 2) L p 6)}|
Then everything is within the (L p) subshell.
Silicon dioxide, SiO2, |{(L p 6 (L s 2 {K s 2}))}Oxygen|Silicon{(({K s 2} L s 2) L p 6)}Silicon|Oxygen{(({K s 2} L s 2) L p 6)}|
Notice that all the electrons from the Sulfur M-shell moved into the Oxygen (L p) shell to fill them. You have three atoms with their L-shells filled. The M-shell has vanished, yet the electrons still "belong" to the Sulfur atom, tying them all together.
_____________
Now, If along the way you guys got totally lost, don't worry, I'm lost too, but this is close. It is going to take getting a handle on the geometries of the electron shells, understanding how the orbits change as energy is pumped in, to really pin any of this stuff down, but this is doable. If you work through the Wiki pages to get the concepts down, you will begin to see how this works.
As always, read the links and make up your own minds.
_____________
Wikipedia tabs opened:
Neutron
http://en.wikipedia.org/wiki/Neutron
Proton
http://en.wikipedia.org/wiki/Proton
Electron
http://en.wikipedia.org/wiki/Electron
Neutrino
http://en.wikipedia.org/wiki/Neutrino
Electron shell
http://en.wikipedia.org/wiki/Electron_shell
Covalent bond
http://en.wikipedia.org/wiki/Covalent_bond
Molecular orbital theory
http://en.wikipedia.org/wiki/Molecular_Orbital_Theory
Hydrogen atom
http://en.wikipedia.org/wiki/Hydrogen-1
Isotopes of hydrogen
http://en.wikipedia.org/wiki/Isotopes_of_hydrogen
Deuterium
http://en.wikipedia.org/wiki/Deuterium
Heavy water
http://en.wikipedia.org/wiki/Heavy_water
Carbon
http://en.wikipedia.org/wiki/Carbon
Nitrogen
http://en.wikipedia.org/wiki/Nitrogen
Oxygen
http://en.wikipedia.org/wiki/Oxygen
Carbon monoxide
http://en.wikipedia.org/wiki/Carbon_monoxide
Sulfur
http://en.wikipedia.org/wiki/Sulphur
Sulfur dioxide
http://en.wikipedia.org/wiki/Sulfur_dioxide
Silicon dioxide
http://en.wikipedia.org/wiki/Silicon_dioxide
Delocalized electron
http://en.wikipedia.org/wiki/Delocalized_electron
Atomic orbital
http://en.wikipedia.org/wiki/Atomic_orbitals
Pauli exclusion principle
http://en.wikipedia.org/wiki/Pauli_exclusion_principle
18-Electron rule
http://en.wikipedia.org/wiki/18-Electron_rule